Image
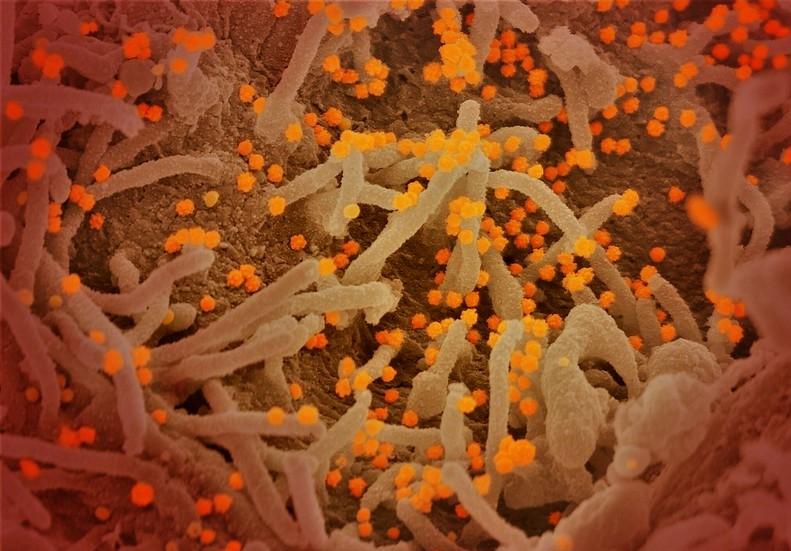
The novel coronavirus captured by a lab at the National Institute of Allergy and Infectious Diseases.
Image
SHUTTERSTOCK
Image

Malheur County.
|
SHUTTERSTOCK
Image

The VA hospital in Southwest Portland/VA Portland Health Care System
Image

Shutterstock
Image
Image

PHOTO BY ENGIN AKYURT ON UNSPLASH
Image
FRITZ LIEDTKE/OREGON HEALTH & SCIENCE UNIVERSITY
Image
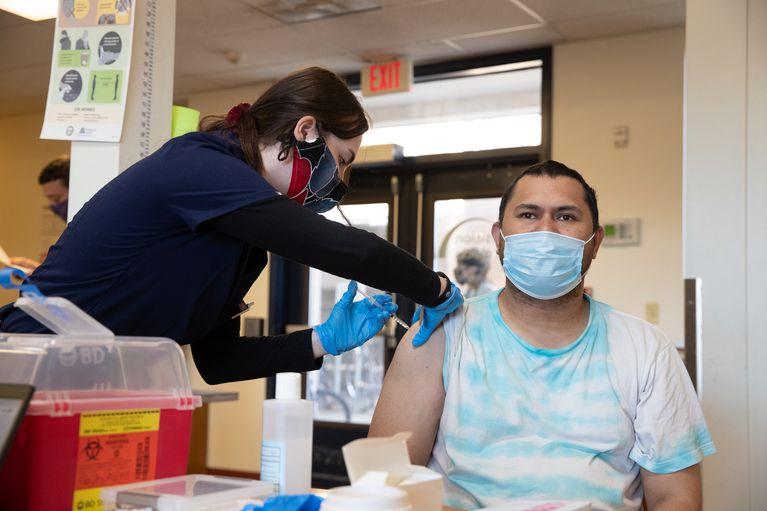
Multnomah County worked to vaccinate people of color and people experiencing homelessness.
|
MOTOYA NAKAMURA/MULTNOMAH COUNTY
Image

Virginia Garcia medical services is trying to increase the vaccination rates of farmworkers.
|
VIRGINIA GARCIA