The Lund Report is offering this coverage for free to better inform the public at this difficult time. But we need your support to help us stretch our resources. Please sign up for a tax-deductible premium subscription or consider making a donation. That is also tax deductible because we're a 501(c)(3) organization.
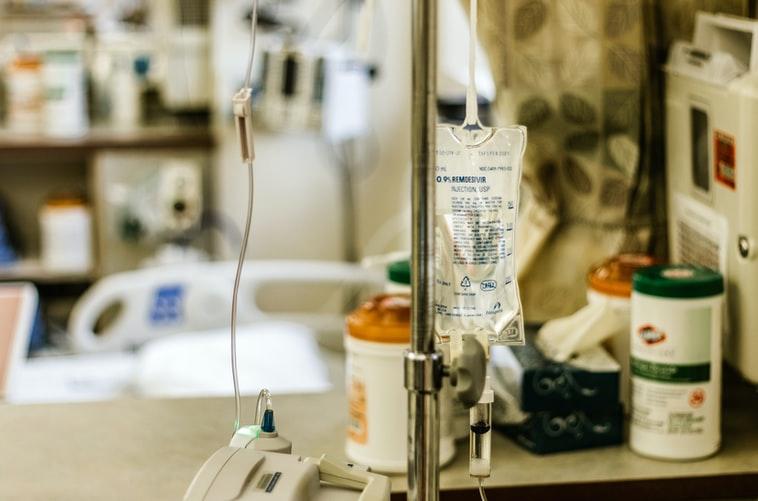
Oregon has received its first shipments of remdesivir, an experimental drug that has been used to treat patients hospitalized with severe COVID-19.
On May 12 and May 15, Oregon received allotments of the drug, which has not been formally approved by the Food and Drug Administration (FDA), but is being used under a federally issued Emergency Use Authorization (EUA).
The EUA allows health professionals to use the drug to treat some severely ill COVID-19 patients who meet clinical criteria. Remdesivir was developed by Gilead Sciences Inc. and has been tested in patients with various diseases, such as Ebola, Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS). According to Gilead’s website, “it is not known if remdesivir is safe and effective for the treatment of COVID-19.”
Preliminary clinical testing by the National Institute for Allergy and Infectious Disease shows that some patients treated with the drug experienced faster recoveries.
“The Oregon Health Authority is committed to distributing Oregon’s allotment of remdesivir to Oregon hospitals for the treatment of patients with severe COVID-19 in accordance with the FDA’s Emergency Use Authorization,” said Dr. Dana Hargunani, Chief Medical Officer. “Because of the experimental nature of the drug, shared decision-making between patients and providers is paramount, and informed consent must be obtained prior to its use.”
Preliminary results, released in April, showed that individuals with advanced lung disease who received the drug recovered 31% faster than patients who did not. The study included 1,063 patients. Differences in the mortality rate were not statistically significant. Full study results have not been published.
Providence St. Vincent and Providence Portland medical centers have been approved for clinical trials of the drug. More than 30 patients have been tested.
“Clinical observation by providers across the board has been that overall clinical improvement and in particular fever resolution, is sped up by the drug’s administration. It has varied by the stage of the patients’ degree of illness at time of presentation. Those findings were confirmed in the recent preliminary data release by Gilead last week – patients were able to discharge 4 to 5 days sooner with drug therapy.” said Dr. Tobias Pusch, an infectious disease physician at Providence.
The allotments received by OHA included enough remdesivir for 80 patients to receive a 10-day treatment course. As of May 16, this is enough to treat all currently hospitalized COVID-19 patients who meet eligibility criteria. OHA anticipates additional allotments in the future, although the amount and timing are not known.
The drug will be distributed to hospitals immediately upon notification of an eligible patient and under specified terms of the EUA.
As of May 16, at 8:30 a.m. there were 56 people hospitalized with confirmed COVID 19. Among those 25 are being treated in an intensive care unit.
Learn more: